In heart attacks, cardiomyocytes, or muscle cells, are attacked. If artificial heart tissue is made, it must follow the structure and function of the heart. The heart is an organ that transmits signals to cells for the regulation of muscle contractions (heart beating). Heart tissue that is lab-grown must have a scaffold that is electrically conductive so it can also do this.
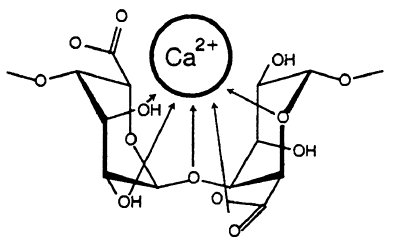
Cardiomyocytes have been shown to grow on alginate or gelatin scaffolds in the past. Alginate is a polysaccharide made up of α-L-guluronic acid and β-D-mannuronic acid. Because alginate is found in seaweeds, its structure is very gel-like and flexible. When alginate interacts with metal ions, such as calcium ions, hydrophilic gels are formed. In terms of structure, both poly guluronic acid and poly mannuronic acid bind to calcium ions. However, poly guluronic acid interacts more strongly to calcium by cross linking, and it also has more hydrogen bonding between the carboxyl groups and OH groups. Alginate structures with a greater amount of poly guluronic acid thus have stronger structures.
| Sodium polymannuronic acid |
 |
| Sodium polyguluronic acid |
The carbon nanotubes had a crosslinked methacrylated gelatin film on it, allowing the hydrogel to be formed. This new hydrogel imitated the Punkinje fibers in the heart that have conduction capabilities. It was also seen that these new cells beat at a faster rate than tissue that contained only gelatin and could use an 85% weaker electric field to beat.
In the future, these nanotube-grown cells will be continually used in cardiac tissue applications, as well as in other organs of the body that require the withstanding of contractions.
http://cen.acs.org/articles/91/web/2013/02/Carbon-Nanotubes-Help-Grow-Beating.html